Applications
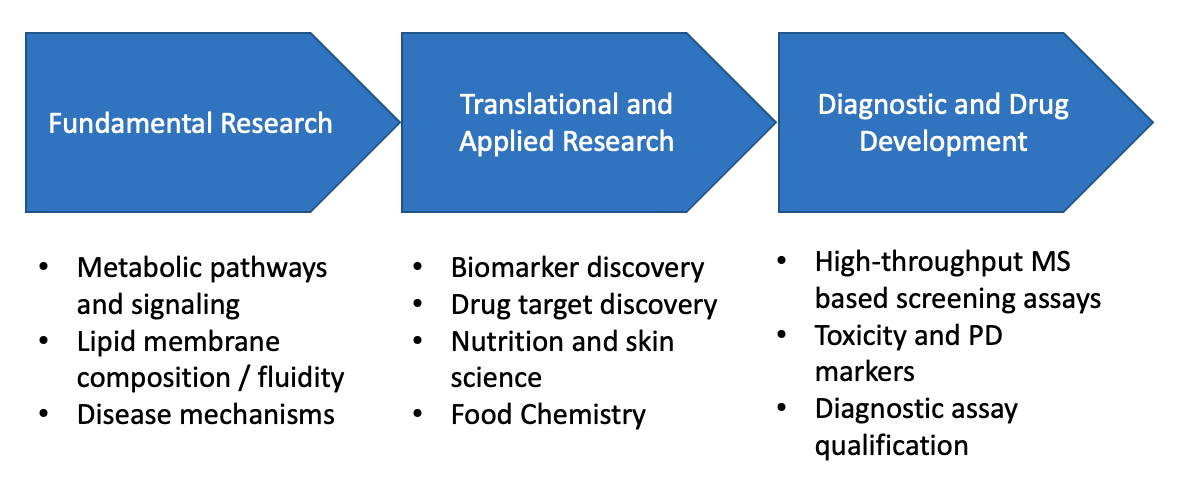
In recent years, the science of lipidomics has emerged as an important tool for fundamental and translational research. The human “lipidome” is composed of almost 200,000 different lipid species, as components of membranes, and functions in signaling, energy storage and anchors for proteins. As such, many disease states are associated with alteration in lipid metabolism, including obesity, cardiovascular, inflammatory , cancer and neurodegenerative disease. Recent progress in pre-analytics and MS-instruments has enabled detection and quantification of many lipid species, thus providing novel insights into the underlying biology.
Selected publications
ELOVL5 Is a Critical and Targetable Fatty Acid Elongase in Prostate Cancer
Margaret M. Centenera et al, Cancer Research, 2021, DOI: 10.1158/0008-5472.CAN-20-2511
Targeted lipidomics analysis of disease models and patient-derived samples was used to identify fatty acid elongation as one of the hallmarks of prostate cancer. This led to the discovery of a key enzyme and possible drug target driving this phenotype – ELOVL5.
Synthesis and fluorine-18 radiolabeling of a phospholipid as a PET imaging agent for prostate cancer
Kim H.Kwan et al, Nuclear Medicine and Biology, 2021, https://doi.org/10.1016/j.nucmedbio.2020.11.007
Lipidomics analysis was used to identify the polyunsaturated phospholipid PC44:12 as a highly (up)regulated androgen-dependent target for PET-tracer development in prostate cancer. The novel tracer was validated in mice models to monitor the distribution of phospholipids in vivo.
Ceramide analog [18F]F-HPA-12 detects sphingolipid disbalance in the brain of Alzheimer’s disease transgenic mice by functioning as a metabolic probe
Simone M. Crivelli et al, Scientific Reports, 2020, https://doi.org/10.1038/s41598-020-76335-4
The authors used Targeted lipidomics to demonstrate significant and specific differences in sphingolipid metabolism in Alzheimer’s mice models, and to validate a novel metabolic probe for ceramide disbalance in the brain.
The role of lipids in determining the air-water interfacial properties of wheat, rye, and oat dough liquor constituents
Frederik Janssen et al, Food Chemistry, 2020, https://doi.org/10.1016/j.foodchem.2020.126565
Targeted lipidomics was used to identify lipids classes in wheat. The study found that predominantly non-polar lipid classes were the main lipid constituents, while lipids played a determining role in the quality of the dough for bread-making.
Human DECR1 is an androgen-repressed survival factor that regulates PUFA oxidation to Protect prostate tumor cells from ferroptosis
Zeyad D Nassar et al, eLife, 2020, DOI: 10.7554/eLife.54166
The authors employed targeted lipidomics to study the effect of a knock-down of a key lipid metabolism enzyme and drug target in prostate cancer – DECR1. The study found that PUFA oxidation promotes prostate cancer cell survival and resistance to androgen receptor targeting therapeutics.
Stearoyl-CoA desaturase-1 impairs the reparative properties of macrophages and microglia in the brain
Jeroen F.J. Bogie et al, JEM, 2020 https://doi.org/10.1084/jem.20191660
The authors started from the observation that a fatty acid desaturase - SCD1 is overexpressed in macrophages that contribute to the immuno-cellular response in neurodegeneration. Targeted lipidomics showed a sustained increase in unsaturated fatty acids in these cells that ultimately resulted in lipid accumulation and a disease-promoting inflammatory phenotype. The study thereby identified SCD-1 as a novel drug target.
The Transfer of Sphingomyelinase Contributes to Drug Resistance in Multiple Myeloma
Sylvia Faict et al, Cancers, 2019 DOI: 10.3390/cancers11121823
The authors employed targeted lipidomics to identify differences in lipid composition of plasma between healthy and Multiple Myeloma patients to identify new targets for overcoming drug resistance. The downregulation of sphingomyelin, driven by acid sphingomyelinase (ASM) was identified as key component responsible for drug resistance, while ASM inhibition increased sensitivity to anti-myeloma drugs.
Evidence for an alternative fatty acid desaturation pathway increasing cancer plasticity
Kim Vriens et al, Nature, 2019, DOI: 10.1038/s41586-019-0904-1
The authors discover that there is an alternative fatty acid desaturation pathway in cancer cells, correlating with the abundance of the unusual fatty acid sapianate, and driven by FASD2. The phospholipid products of this fatty acid are incorporated into cell membranes, as determined by targeted lipidomics, affecting membrane plasticity and resistance to lipid peroxidation.
Sustained SREBP-1-dependent lipogenesis as a key mediator of resistance to BRAF-targeted therapy
Ali Talebi et al, Nature Communications, 2018, https://doi.org/10.1038/s41467-018-04664-0
The authors found that resistance to BRAF-targeted therapy is correlated to sustained lipogenesis via fatty acid biosynthesis driven by SREBP-1. Targeted lipidomics analysis demonstrated that this results in increased incorporation of saturated and mono-unsaturated phospholipids in cell membranes, thereby protecting melanoma from ROS-induced lipid peroxidation, and identifying SREBP-1 as a novel drug target.
Cell density-dependent ferroptosis in breast cancer is induced by accumulation of polyunsaturated fatty acid-enriched triacylglycerides
Elena Panzilius et al, BioRxiv, 2019, https://doi.org/10.1101/417949
The authors start for the observation that cell density plays a critical role in determining sensitivity to ferroptosis in breast cancer. Targeted lipidomics was then used to demonstrate that cells at low density accumulate triacyl-glycerides with PUFA fatty acids, which may serve a protective role against ROS-induced lipid peroxidation, but in turn provides a positive signal for ferroptosis.